Endotoxin testing System (Multiple Samples) Endosafe® nexgen-MCS™
Description
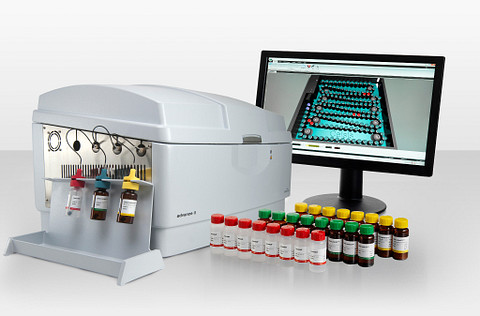
The Endosafe® nexgen-MCS™ is a high-throughput endotoxin testing system designed for fast, accurate, and compliant results. Delivering quantitative endotoxin data in just 15 minutes, the system can test up to five samples simultaneously, with each sample running independently for true random-access flexibility.
Using FDA-licensed Endosafe®-PTS™ cartridges, the system ensures consistent performance and full compliance with USP/EP BET guidelines. It supports four cartridge sensitivity levels—0.005, 0.01, 0.05, and 0.10 EU/mL—to accommodate a wide range of testing requirements.
Built for regulated environments, the nexgen-MCS™ is fully 21 CFR Part 11 compliant, making it ideal for reliable, high-volume endotoxin testing in modern laboratories.



Reviews
There are no reviews yet.