
Rapid Kinetic Endotoxin Testing
Description
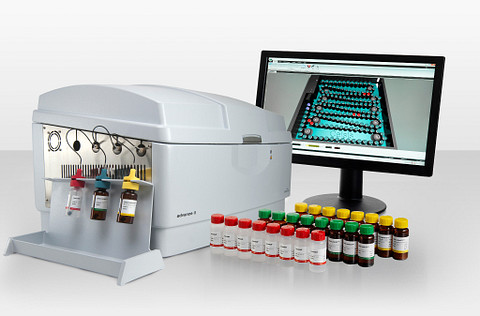
The portable Rapid Kinetic Endotoxin Testing System delivers fast, accurate, and fully quantitative endotoxin results in just 15 minutes. Each unit comes equipped with a dedicated reader and FDA-licensed LAL cartridges, ensuring dependable performance in critical testing environments.
Built for compliance, the system meets 21 CFR Part 11 requirements and fully aligns with harmonized BET standards outlined in USP <85> and EP <2.6.14>. Every cartridge performs an automatic duplicate sample and positive product control, eliminating manual steps and guaranteeing consistent, regulatory-approved results.
All quantitative data can be seamlessly exported to a central PC for efficient tracking, trending, and documentation. To support diverse testing needs, cartridges are available in three sensitivity levels: 0.01, 0.05, and 0.005 EU/mL.



Reviews
There are no reviews yet.